Ajoure
PRODUCT INFORMATION
NAME
AJOURE (with lidocaine)
Packaging
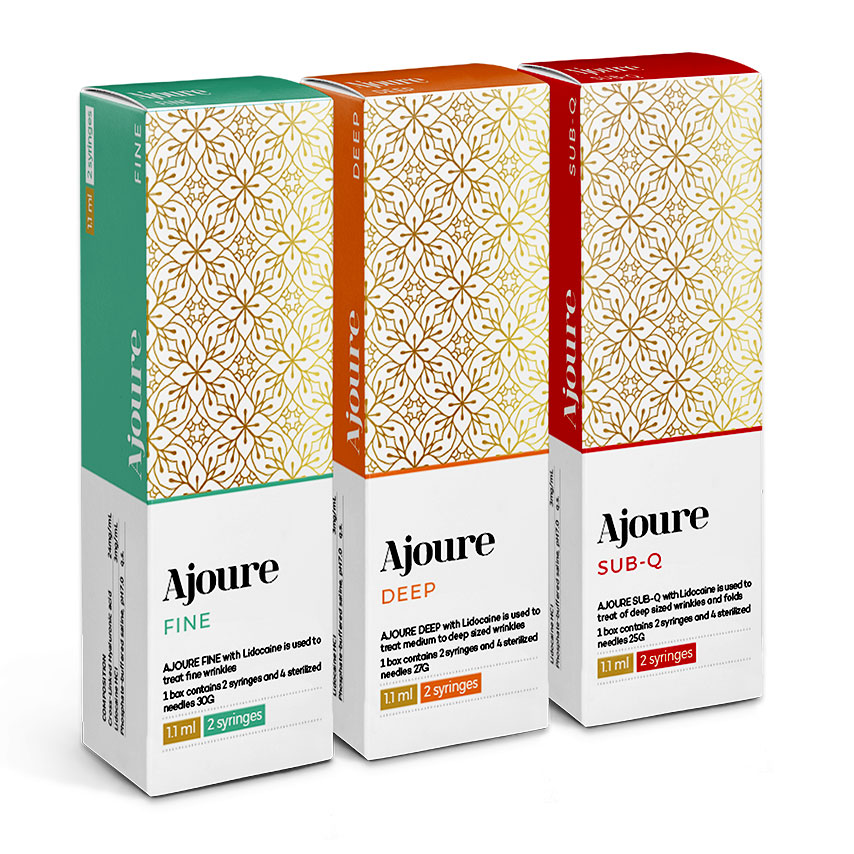
Ajoure with lidocaine 1.1 ml*2 syringe
How to use
Properly designed syringe and plunger design maintains even pressure, providing a comfortable administration of the drug.
Storage Condition
Store at 2-25°C. Do not freeze, keep out of direct sunlight.
VENDOR
T.A Bio Tech Co., Ltd.
PRODUCT INTRODUCTION
Ajoure is a filler with a stable gel that was made by precision cross-linking technology.
Performance: Hyaluronic acid is a high molecular substance composed of repeated disaccharides of N-acetyl glucosamin and glucuronic acid. Ajoure has realized a stable gel through precision cross-linking technology. The stable structure of hyaluronic acid gel fills the space, supports the structure, protects the cell, replenishes the volume of wrinkled skin and keeps it natural.
Safety: The active ingredient used in Ajoure is non-animal. It is a human-adapted component، contains high-purity hyaluronic acid and naturally decomposed over time. Hyaluronic acid cross-linking gel can be corrected through hyaluronidase when a problem occurs. It is safe by providing the level of endotoxin and crosslinking agent below the detection limit through strict manufacturing process management.
Usability: Precisely designed handles and push rods keep the pressure at the time of injection evenly،enabling stable treatment. The monophasic structure is a regular and stable gel structure، and it is a specialized technology applied to Ajoure that realizes natural volume and soft injection feeling.
Pain relief: Ajoure contains Lidocaine to improve pain-related discomfort during the treatment.
Storage Condition: Store at 2-25°C. Do not freeze, keep out of direct sunlight.
How to use: Should be performed by a certified professional.
Packaging: 1.1 ml * 2 syringe.
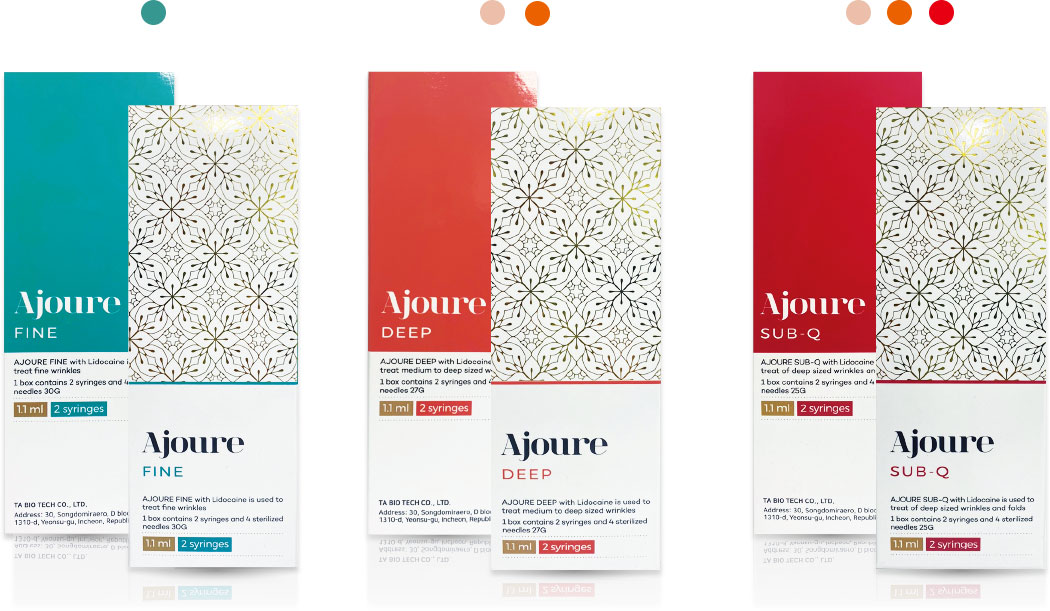
| Product Name | FINE | DEEP | SUB-Q |
|---|---|---|---|
| Injection Site | Superficial to mid dermis | Mid to deep dermis | Mid to deep dermis subcutaneous layer |
| HA Concentration | 24 mg/ml | ||
| Visco-elasticity | Low | Mid | High |
| Duration | 6-9 months | 9-18 months | 12-24 months |
| Packaging unit | 30G*2 | 27G*2 | 25G*2 |
| Injection Area | All facial wrinkles, lip augmentation | Nasolabial folds, forehead, marionette lines and face lifting | Facial contouring, chin and chick augmentation |
| Syringe Volume | 1.1 ml x 2 syringes | ||
| Lidocaine | 0,3% | ||
| Shelf Life | 24 months | ||
| Storage | Store in 2-25°C. Protect form direct sunlight and freezing | ||
DETAILED

Ajoure
FINE | DEEP | SUB-Q
| Product Name | FINE | DEEP | SUB-Q |
|---|---|---|---|
| Injection Site | Superficial to mid dermis | Mid to deep dermis | Mid to deep dermis subcutaneous layer |
| HA Concentration | 24 mg/ml | ||
| Visco-elasticity | Low | Mid | High |
| Duration | 6-9 months | 9-18 months | 12-24 months |
| Packaging unit | 30G*2 | 27G*2 | 25G*2 |
| Injection Area | All facial wrinkles, lip augmentation | Nasolabial folds, forehead, marionette lines and face lifting | Facial contouring, chin and chick augmentation |
| Syringe Volume | 1.1 ml x 2 syringes | ||
| Lidocaine | 0,3% | ||
| Shelf Life | 24 months | ||
| Storage | Store in 2-25°C. Protect form direct sunlight and freezing | ||
*The durations are depends on aage, lifestyle and the skill of a practitioner.

DISPOSABLE MEDICAL DEVICE

100% Korean High Quality Raw materials
AJOURE is manufactured using 100% authentic Korean high-quality hyaluronic acid, produced at the one of the biggest pharmaceutical factories in South Korea.
Finished product complies with European Pharmacopoeia on the endotoxin level.
| Test Items | Acceptance Criteria | Results |
|---|---|---|
| Endotoxin | Not more than 12.5 EU/ml | Less than 0.5 EU/ml |
High purification Process
Only AJOURE's unique manufacturing technology enables high elasticity with lower amount of cross-linking agent and residual BDDE
| Test Items | Acceptance Criteria | Results |
|---|---|---|
| BDDE | ≤2ppm(NMT 2ppm) | Not detected |

High modification Low cross-linking

Moderate modification Moderate cross-linking

Low modification High cross-linking
High Elasticity & Cohesiveness
No swelling, no break during injection, maintaining original shape and natural look


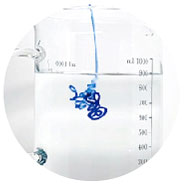

Keep shape after injection




Not easily broken by water

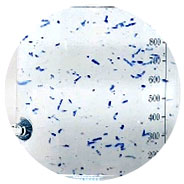


| Test Items | Acceptance Criteria | Results |
|---|---|---|
| Viscosity (Apparent viscosity, 50Hz) | 131OOcp~17OOOcp | 16000cp |
High viscoelasticity and cohesiveness, but very comfortable injection force


| Test Items | Acceptance Criteria | Results |
|---|---|---|
| Extrusion force | Not more than 25N | 10N |

















FINE
1 box contains 2 syringes and 4 sterilized needles 3OG
1.1 ml
2 syringes
DEEP
1 box contains 2 syringes and 4 sterilized needles 27G
1.1 ml
2 syringes
SUB-Q
1 box contains 2 syringes and 4 sterilized needles 25G
1.1 ml
2 syringes
DISPOSABLE MEDICAL DEVICE
Before-after photos
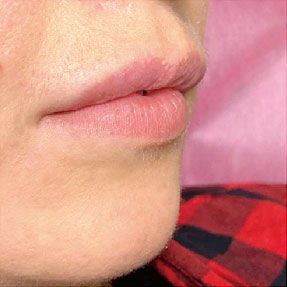
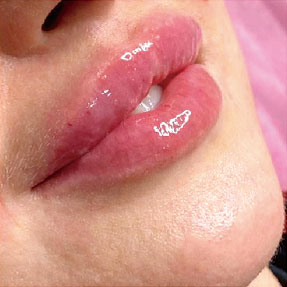
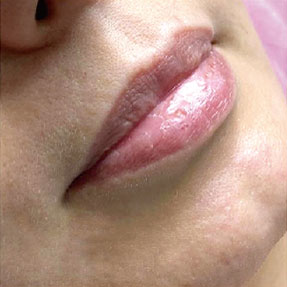
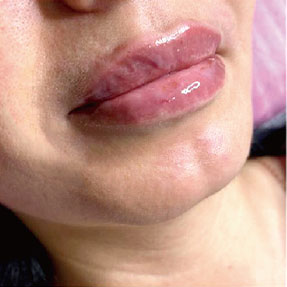
FINE
DEEP
SUB-Q
- Hyaluronic Acid
- Dermal Filler
- 24 mg/ml
- Lidocaine 3 mg/ml
- High HA concentration
- Longer Duration
- Safety Raw materials
- No residual BDDE
- High Elasticity
COMPOSITION
Cross-Linked Hyaluronic acid
Lidocaine HCI
Phosphate-buffered saline, pH7.0
24mg/ml
3mg/mL
q.s.